Aripiprazole 1mg/ml Oral Solution
Therapeutic indications include:
- Treatment of schizophrenia in adults and in adolescents aged 15 years and older.
- Treatment of moderate to severe manic episodes in Bipolar I Disorder and for the prevention of a new manic episode in adults who experienced predominantly manic episodes and whose manic episodes responded to aripiprazole treatment.
- Treatment up to 12 weeks of moderate to severe manic episodes in Bipolar I Disorder in adolescents aged 13 years and older1
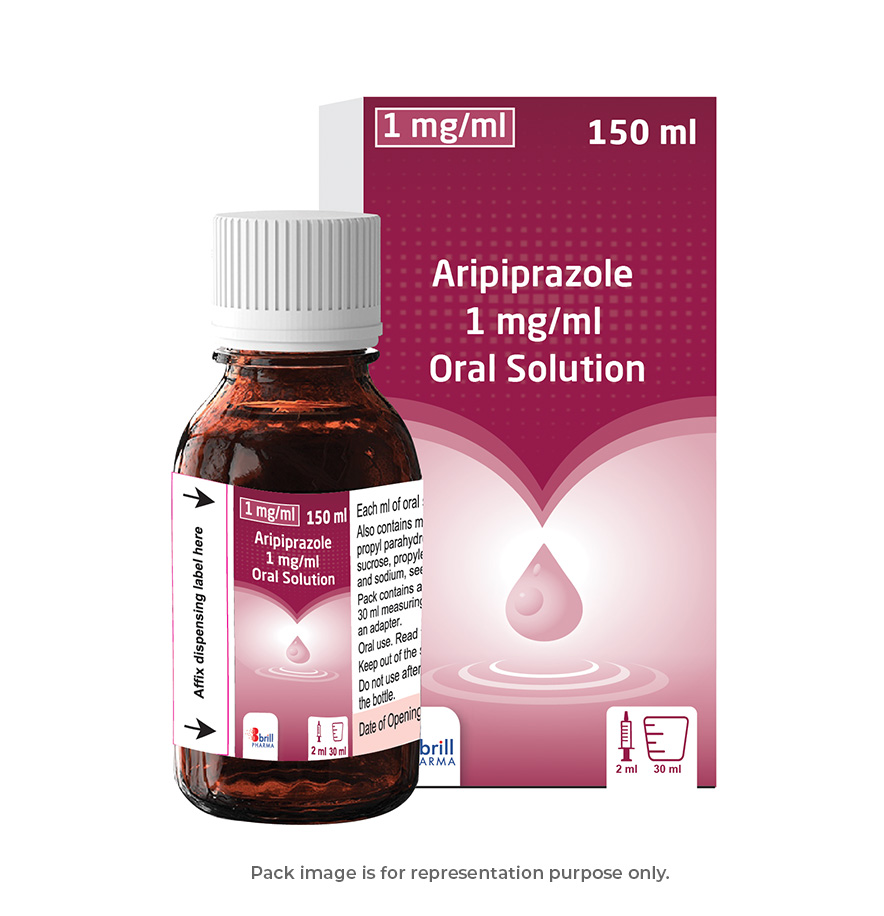
| Strength | 1mg/ ml |
| Pack Size | 150 ml |
| Measuring Device | A calibrated polypropylene 30ml measuring cup (graduated at every 2.5ml equivalent to 2.5mg) and a 2ml oral syringe with adaptor (graduated at every 0.5 ml equivalent to 0.5mg) |
| Regulatory Status | POM |
| Flavour | Orange |
| Suitable for Vegetarians | Yes |
| GTIN | 5060712420072 |
| PIP code | 1244441 |
Product information and Risk materials
| Summary of Product Characteristics (SPC) | Download |
| Patient Leaflet (PIL) | Download |
| Risk Materials | – |

Manufacturing
Our quality and safety teams monitor and analyse each stage of the production and distribution process to make sure our products are of the highest quality. The application of Good Manufacturing Practice (GMP) means that we have the appropriate standards and procedures in place to help us achieve this.
How to report adverse events
Adverse events should be reported.
If you are based in the UK, reporting forms and information can be found at: https://yellowcard.mhra.gov.uk.
Adverse events should also be reported to Brill Pharma
Tel: 0044 (0) 1582 545500 Email: info@brillpharma.co.uk
If you are based outside of the UK, please report adverse events to Brill Pharma using the contact details provided above.
Have a question?
If you have a question about Brill Pharma or any products in our range please get in touch
Tel: 0044 (0) 1582 545500 Email: info@brillpharma.co.uk
